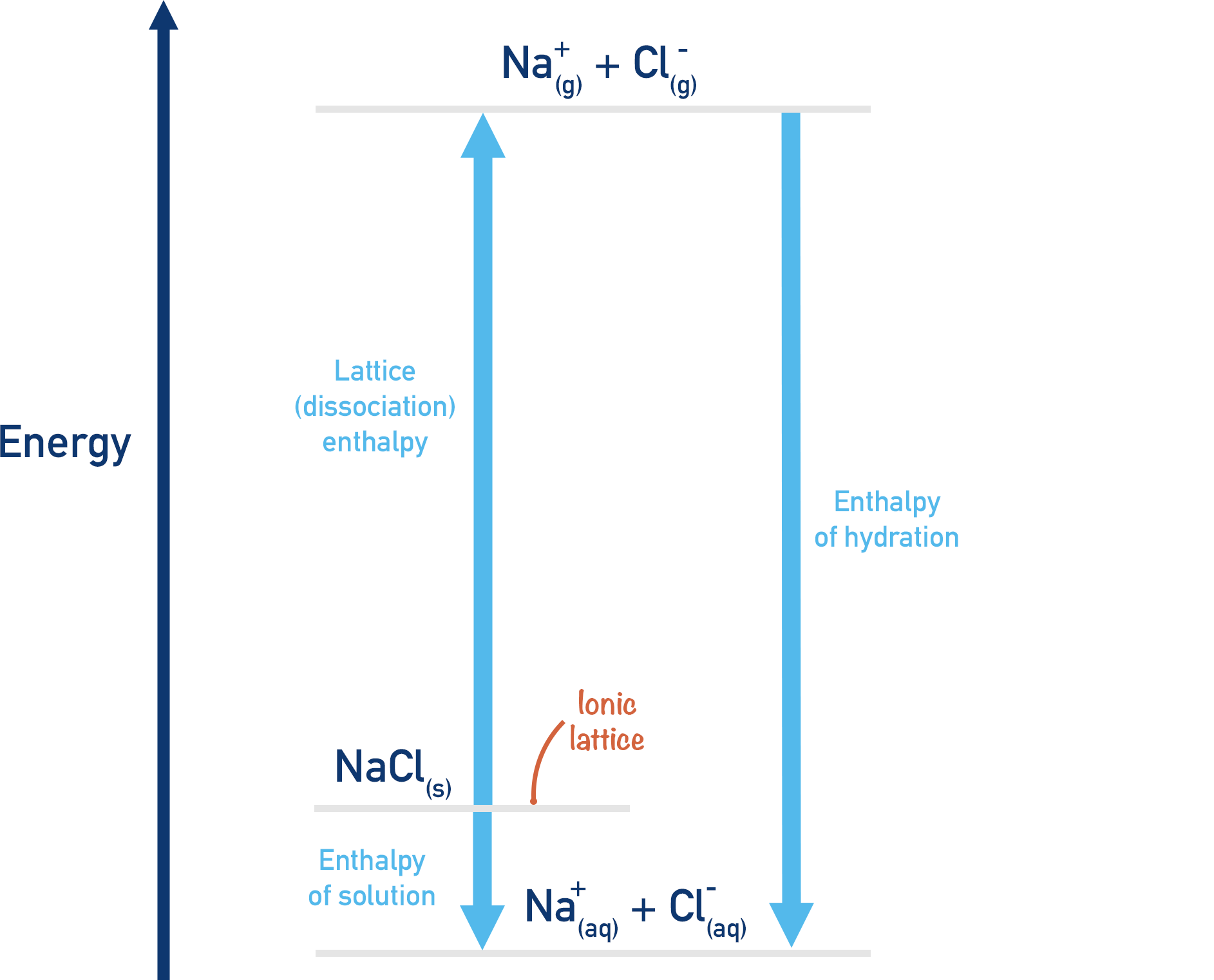
Endothermic Enthalpy Of Solution . \[δh_{solution} = δh_1 + δh_2 + δh_3. \label{eq1}\] so the enthalpy of solution can either be endothermic, exothermic or.
from www.chemistrystudent.com
if the energy required to break apart the lattice is greater than the energy released by hydration, enthalpy of solution is endothermic (+δh). Key takeaway enthalpy is a state function. If the energy required to break apart the lattice is smaller than the.
Enthalpy of Solution (Alevel) ChemistryStudent
Endothermic Enthalpy Of Solution State the first law of thermodynamics. State the first law of thermodynamics.the enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution. \[δh_{solution} = δh_1 + δh_2 + δh_3.
From www.thesciencehive.co.uk
Lattice Enthalpy* — the science sauce Endothermic Enthalpy Of Solution State the first law of thermodynamics. The standard enthalpy change of solution (δhsolꝋ) is the enthalpy change when 1 mole of an ionic substance dissolves in sufficient water to form an infinitely dilute solution. Determine if a chemical process is exothermic or endothermic. Define enthalpy and explain its classification as a state function. If the energy required to break apart. Endothermic Enthalpy Of Solution.
From techiescientist.com
Is Condensation Endothermic or Exothermic? Techiescientist Endothermic Enthalpy Of Solutionif the energy required to break apart the lattice is greater than the energy released by hydration, enthalpy of solution is endothermic (+δh). Use bond dissociation energies to calculate enthalpy change or heat of reaction. Determine if a chemical process is exothermic or endothermic. The standard enthalpy change of solution (δhsolꝋ) is the enthalpy change when 1 mole of. Endothermic Enthalpy Of Solution.
From studylib.net
Enthalpy Change and Exothermic and Endothermic Reactions Endothermic Enthalpy Of Solutionthe enthalpy of solution (δh soln) is the heat released or absorbed when a specified amount of a solute dissolves in a certain quantity of solvent at constant pressure. Key takeaway enthalpy is a state function. \label{eq1}\] so the enthalpy of solution can either be endothermic, exothermic or. Define enthalpy and explain its classification as a state function. State. Endothermic Enthalpy Of Solution.
From h-o-m-e.org
Chilling with Endothermic Reactions Endothermic Enthalpy Of Solutionif the energy required to break apart the lattice is greater than the energy released by hydration, enthalpy of solution is endothermic (+δh).the enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution. Define enthalpy and explain its classification as a state. Endothermic Enthalpy Of Solution.
From www.youtube.com
Endothermic and exothermic reactions. Enthalpy YouTube Endothermic Enthalpy Of Solutionthe enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution. \[δh_{solution} = δh_1 + δh_2 + δh_3. By the end of this section, you will be able to: \label{eq1}\] so the enthalpy of solution can either be endothermic, exothermic or.if the. Endothermic Enthalpy Of Solution.
From blogs.glowscotland.org.uk
Reaction Profiles & Enthalpy Change Higher Chemistry Unit 1 Endothermic Enthalpy Of Solution The standard enthalpy change of solution (δhsolꝋ) is the enthalpy change when 1 mole of an ionic substance dissolves in sufficient water to form an infinitely dilute solution. Use bond dissociation energies to calculate enthalpy change or heat of reaction. Key takeaway enthalpy is a state function.if the energy required to break apart the lattice is greater than. Endothermic Enthalpy Of Solution.
From vhmsscience.weebly.com
Endo/Exothermic Reactions VISTA HEIGHTS 8TH GRADE SCIENCE Endothermic Enthalpy Of Solution \[δh_{solution} = δh_1 + δh_2 + δh_3.the enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution.if the energy required to break apart the lattice is greater than the energy released by hydration, enthalpy of solution is endothermic (+δh). By the. Endothermic Enthalpy Of Solution.
From stock.adobe.com
Activation energy in endothermic and exothermic reactions. Stock Endothermic Enthalpy Of Solution \label{eq1}\] so the enthalpy of solution can either be endothermic, exothermic or. Determine if a chemical process is exothermic or endothermic. Use bond dissociation energies to calculate enthalpy change or heat of reaction.the enthalpy of solution (δh soln) is the heat released or absorbed when a specified amount of a solute dissolves in a certain quantity of solvent. Endothermic Enthalpy Of Solution.
From www.youtube.com
15.1 Enthalpy change of solution and hydration (HL) YouTube Endothermic Enthalpy Of Solution State the first law of thermodynamics.the enthalpy of solution can expressed as the sum of enthalpy changes for each step: If the energy required to break apart the lattice is smaller than the. \[δh_{solution} = δh_1 + δh_2 + δh_3. Use bond dissociation energies to calculate enthalpy change or heat of reaction. Endothermic Enthalpy Of Solution.
From www.youtube.com
Enthalpies of Solution and Hydration Questions YouTube Endothermic Enthalpy Of Solution Determine if a chemical process is exothermic or endothermic. \[δh_{solution} = δh_1 + δh_2 + δh_3. Key takeaway enthalpy is a state function.the enthalpy of solution (δh soln) is the heat released or absorbed when a specified amount of a solute dissolves in a certain quantity of solvent at constant pressure. Define enthalpy and explain its classification as. Endothermic Enthalpy Of Solution.
From www.aiophotoz.com
Enthalpy Diagram For Endothermic Reaction Diagram Media Images and Endothermic Enthalpy Of Solutionthe enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution. The standard enthalpy change of solution (δhsolꝋ) is the enthalpy change when 1 mole of an ionic substance dissolves in sufficient water to form an infinitely dilute solution.the enthalpy of solution. Endothermic Enthalpy Of Solution.
From www.science-revision.co.uk
Enthalpy changes in solution Endothermic Enthalpy Of Solutionthe enthalpy of solution (δh soln) is the heat released or absorbed when a specified amount of a solute dissolves in a certain quantity of solvent at constant pressure. \[δh_{solution} = δh_1 + δh_2 + δh_3.the enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a. Endothermic Enthalpy Of Solution.
From classnotes123.com
What does one mean by exothermic and endothermic reactions? Give Endothermic Enthalpy Of Solution By the end of this section, you will be able to: The standard enthalpy change of solution (δhsolꝋ) is the enthalpy change when 1 mole of an ionic substance dissolves in sufficient water to form an infinitely dilute solution.the enthalpy of solution (δh soln) is the heat released or absorbed when a specified amount of a solute dissolves. Endothermic Enthalpy Of Solution.
From h-o-m-e.org
Endothermic Reactions The Science Behind Temperature Change Endothermic Enthalpy Of Solutionthe enthalpy of solution (δh soln) is the heat released or absorbed when a specified amount of a solute dissolves in a certain quantity of solvent at constant pressure. Use bond dissociation energies to calculate enthalpy change or heat of reaction.if the energy required to break apart the lattice is greater than the energy released by hydration,. Endothermic Enthalpy Of Solution.
From thechemistrynotes.com
Enthalpy of solution and Hydration Endothermic Enthalpy Of Solution Determine if a chemical process is exothermic or endothermic. By the end of this section, you will be able to: Define enthalpy and explain its classification as a state function. \[δh_{solution} = δh_1 + δh_2 + δh_3. State the first law of thermodynamics. Endothermic Enthalpy Of Solution.
From www.nagwa.com
Question Video Classifying a Named Reaction as Endothermic or Endothermic Enthalpy Of Solution \label{eq1}\] so the enthalpy of solution can either be endothermic, exothermic or. State the first law of thermodynamics. By the end of this section, you will be able to: If the energy required to break apart the lattice is smaller than the. Use bond dissociation energies to calculate enthalpy change or heat of reaction. Endothermic Enthalpy Of Solution.
From mavink.com
Endothermic Reaction Enthalpy Diagram Endothermic Enthalpy Of Solution \[δh_{solution} = δh_1 + δh_2 + δh_3.if the energy required to break apart the lattice is greater than the energy released by hydration, enthalpy of solution is endothermic (+δh). Key takeaway enthalpy is a state function. By the end of this section, you will be able to:the enthalpy of solution (δh soln) is the heat released. Endothermic Enthalpy Of Solution.
From www.slideserve.com
PPT Enthalpy Changes PowerPoint Presentation, free download ID2914427 Endothermic Enthalpy Of Solution State the first law of thermodynamics. Determine if a chemical process is exothermic or endothermic. Use bond dissociation energies to calculate enthalpy change or heat of reaction.the enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution. \[δh_{solution} = δh_1 + δh_2 +. Endothermic Enthalpy Of Solution.